Where Are the Lab-Grown Replacement Organs?
A futuristic rendering of a scientist holding a pair of lungs in a glass dish.
The headline blared from newspapers all the way back in 2006: "First Lab-Grown Organs Implanted in Humans!" A team from Wake Forest University had biopsied cells from the bladders of patients with spina bifida and used them to create brand new full-size bladders, which they then implanted. Although the bladders had to be emptied via catheter, they were still functioning a few years after implantation, and the public grew confident that doctors had climbed an intermediary step on the way to the medicine of science fiction. Ten years later, though, more than 20 people a day are still dying while waiting for an organ transplant, which leads to a simple question: Where are our fake organs?
"We can make small organs and tissues but we can't make larger ones."
Not coming anytime soon, unfortunately. The company that was created to transition Wake Forest's bladders to the market failed. And while there are a few simple bioengineered skins and cartilages already on the market, they are hardly identical to the real thing. Something like a liver could take another 20 to 25 years, says Shay Soker, professor at Wake Forest's Institute for Regenerative Medicine. "The first barrier is the technology: We can make small organs and tissues but we can't make larger ones," he says. "Also there are several cell types or functions that you can reliably make from stem cells, but not all of them, so the technology of stem cells has to catch up with what the body can do." Finally, he says, you have support the new organ inside the body, providing it with a circulatory and nervous system and integrating it with the immune system.
While these are all challenging problems, circulation appears to be the most intractable. "Tissue's not able to survive if the cells don't have oxygen, and the bigger it gets, the more complex vasculature you need to keep that alive," says Chiara Ghezzi, research professor in the Tufts University Department of Biomedical Engineering. "Vasculature is highly organized in the body. It has a hierarchical structure, with different branches that have different roles depending on where they are." So far, she says, researchers have had trouble scaling up from capillaries to larger vessels that could be grafted onto blood vessels in a patient's body.
"The FDA is still getting its hands and minds around the field of tissue engineering."
Last, but hardly least, is the question of FDA approval. Lab-grown organs are neither drugs nor medical devices, and the agency is not set up to quickly or easily approve new technologies that don't fit into current categories. "The FDA is still getting its hands and minds around the field of tissue engineering," says Soker. "They were not used to that… so it requires the regulatory and financial federal agencies to really help and support these initiatives."
A pencil eraser-size model of the human brain is now being used for drug development and research.
If all of this sounds discouraging, it's worth mentioning some of the incredible progress the field has made since the first strides toward lab-grown organs began nearly 30 years ago: Though full-size replacement organs are still decades away, many labs have diverted their resources into what they consider an intermediate step, developing miniature organs and systems that can be used for drug development and research. This platform will yield more relevant results (Imagine! Testing cardiovascular drugs on an actual human heart!) and require the deaths of far fewer animals. And it's already here: Two years ago, scientists at Ohio State University developed a pencil eraser-size model of the human brain they intend to use for this exact purpose.
Perhaps the most exciting line of research these days is one that at first doesn't seem to have anything to do with bioengineered organs at all. Along with his colleagues, Chandan Sen, Director of the Center for Regenerative Medicine and Cell-based Therapies at Ohio State University, has developed a nanoscale chip that can turn any cell in the body into any other kind of cell—reverting fully differentiated adult cells into, essentially, stem cells, which can then grow into any tissue you want. Sen has used his chip to reprogram skin cells in the bodies of mice into neurons to help them recover from strokes, and blood vessels to save severe leg injuries. "There's this concept of a bioreactor, where you convince an organ to grow outside the body. They're getting more and more sophisticated over time. But to my mind it will never match the sophistication or complexity of the human body," Sen says. "I believe that in order to have an organ that behaves the way you want it to in the live body, you must use the body itself as a bioreactor, not a bunch of electronic gadgetry." There you have it, the next step in artificial organ manufacture is as crazy as it is intuitive: Grow it back where it was in the first place.
A robot server, controlled remotely by a disabled worker, delivers drinks to patrons at the DAWN cafe in Tokyo.
A sleek, four-foot tall white robot glides across a cafe storefront in Tokyo’s Nihonbashi district, holding a two-tiered serving tray full of tea sandwiches and pastries. The cafe’s patrons smile and say thanks as they take the tray—but it’s not the robot they’re thanking. Instead, the patrons are talking to the person controlling the robot—a restaurant employee who operates the avatar from the comfort of their home.
It’s a typical scene at DAWN, short for Diverse Avatar Working Network—a cafe that launched in Tokyo six years ago as an experimental pop-up and quickly became an overnight success. Today, the cafe is a permanent fixture in Nihonbashi, staffing roughly 60 remote workers who control the robots remotely and communicate to customers via a built-in microphone.
More than just a creative idea, however, DAWN is being hailed as a life-changing opportunity. The workers who control the robots remotely (known as “pilots”) all have disabilities that limit their ability to move around freely and travel outside their homes. Worldwide, an estimated 16 percent of the global population lives with a significant disability—and according to the World Health Organization, these disabilities give rise to other problems, such as exclusion from education, unemployment, and poverty.
These are all problems that Kentaro Yoshifuji, founder and CEO of Ory Laboratory, which supplies the robot servers at DAWN, is looking to correct. Yoshifuji, who was bedridden for several years in high school due to an undisclosed health problem, launched the company to help enable people who are house-bound or bedridden to more fully participate in society, as well as end the loneliness, isolation, and feelings of worthlessness that can sometimes go hand-in-hand with being disabled.
“It’s heartbreaking to think that [people with disabilities] feel they are a burden to society, or that they fear their families suffer by caring for them,” said Yoshifuji in an interview in 2020. “We are dedicating ourselves to providing workable, technology-based solutions. That is our purpose.”
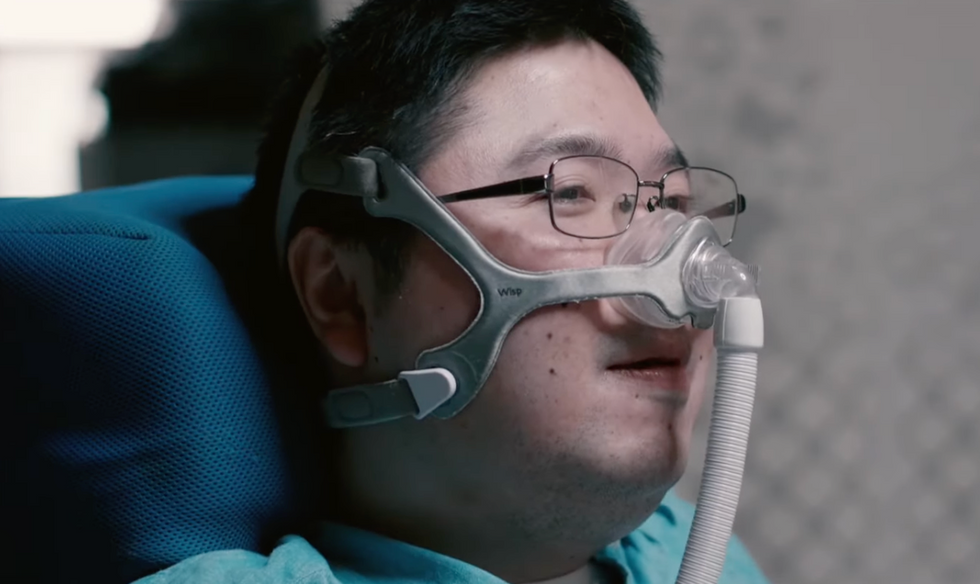
Shota, Kuwahara, a DAWN employee with muscular dystrophy, agrees. "There are many difficulties in my daily life, but I believe my life has a purpose and is not being wasted," he says. "Being useful, able to help other people, even feeling needed by others, is so motivational."
A woman receives a mammogram, which can detect the presence of tumors in a patient's breast.
When a patient is diagnosed with early-stage breast cancer, having surgery to remove the tumor is considered the standard of care. But what happens when a patient can’t have surgery?
Whether it’s due to high blood pressure, advanced age, heart issues, or other reasons, some breast cancer patients don’t qualify for a lumpectomy—one of the most common treatment options for early-stage breast cancer. A lumpectomy surgically removes the tumor while keeping the patient’s breast intact, while a mastectomy removes the entire breast and nearby lymph nodes.
Fortunately, a new technique called cryoablation is now available for breast cancer patients who either aren’t candidates for surgery or don’t feel comfortable undergoing a surgical procedure. With cryoablation, doctors use an ultrasound or CT scan to locate any tumors inside the patient’s breast. They then insert small, needle-like probes into the patient's breast which create an “ice ball” that surrounds the tumor and kills the cancer cells.
Cryoablation has been used for decades to treat cancers of the kidneys and liver—but only in the past few years have doctors been able to use the procedure to treat breast cancer patients. And while clinical trials have shown that cryoablation works for tumors smaller than 1.5 centimeters, a recent clinical trial at Memorial Sloan Kettering Cancer Center in New York has shown that it can work for larger tumors, too.
In this study, doctors performed cryoablation on patients whose tumors were, on average, 2.5 centimeters. The cryoablation procedure lasted for about 30 minutes, and patients were able to go home on the same day following treatment. Doctors then followed up with the patients after 16 months. In the follow-up, doctors found the recurrence rate for tumors after using cryoablation was only 10 percent.
For patients who don’t qualify for surgery, radiation and hormonal therapy is typically used to treat tumors. However, said Yolanda Brice, M.D., an interventional radiologist at Memorial Sloan Kettering Cancer Center, “when treated with only radiation and hormonal therapy, the tumors will eventually return.” Cryotherapy, Brice said, could be a more effective way to treat cancer for patients who can’t have surgery.
“The fact that we only saw a 10 percent recurrence rate in our study is incredibly promising,” she said.


