Genetic Testing Companies Are Facing a Racial Bias Problem in Disease Risk Tests

The large genetic studies underlying certain disease risk tests have primarily been done in populations of European ancestry, limiting their accuracy.
Earlier this year, California-based Ambry Genetics announced that it was discontinuing a test meant to estimate a person's risk of developing prostate or breast cancer. The test looks for variations in a person's DNA that are known to be associated with these cancers.
Known as a polygenic risk score, this type of test adds up the effects of variants in many genes — often in the dozens or hundreds — and calculates a person's risk of developing a particular health condition compared to other people. In this way, polygenic risk scores are different from traditional genetic tests that look for mutations in single genes, such as BRCA1 and BRCA2, which raise the risk of breast cancer.
Traditional genetic tests look for mutations that are relatively rare in the general population but have a large impact on a person's disease risk, like BRCA1 and BRCA2. By contrast, polygenic risk scores scan for more common genetic variants that, on their own, have a small effect on risk. Added together, however, they can raise a person's risk for developing disease.
These scores could become a part of routine healthcare in the next few years. Researchers are developing polygenic risk scores for cancer, heart, disease, diabetes and even depression. Before they can be rolled out widely, they'll have to overcome a key limitation: racial bias.
"The issue with these polygenic risk scores is that the scientific studies which they're based on have primarily been done in individuals of European ancestry," says Sara Riordan, president of the National Society of Genetics Counselors. These scores are calculated by comparing the genetic data of people with and without a particular disease. To make these scores accurate, researchers need genetic data from tens or hundreds of thousands of people.
Myriad's old test would have shown that a Black woman had twice as high of a risk for breast cancer compared to the average woman even if she was at low or average risk.
A 2018 analysis found that 78% of participants included in such large genetic studies, known as genome-wide association studies, were of European descent. That's a problem, because certain disease-associated genetic variants don't appear equally across different racial and ethnic groups. For example, a particular variant in the TTR gene, known as V1221, occurs more frequently in people of African descent. In recent years, the variant has been found in 3 to 4 percent of individuals of African ancestry in the United States. Mutations in this gene can cause protein to build up in the heart, leading to a higher risk of heart failure. A polygenic risk score for heart disease based on genetic data from mostly white people likely wouldn't give accurate risk information to African Americans.
Accuracy in genetic testing matters because such polygenic risk scores could help patients and their doctors make better decisions about their healthcare.
For instance, if a polygenic risk score determines that a woman is at higher-than-average risk of breast cancer, her doctor might recommend more frequent mammograms — X-rays that take a picture of the breast. Or, if a risk score reveals that a patient is more predisposed to heart attack, a doctor might prescribe preventive statins, a type of cholesterol-lowering drug.
"Let's be clear, these are not diagnostic tools," says Alicia Martin, a population and statistical geneticist at the Broad Institute of MIT and Harvard. "We can't use a polygenic score to say you will or will not get breast cancer or have a heart attack."
But combining a patient's polygenic risk score with other factors that affect disease risk — like age, weight, medication use or smoking status — may provide a better sense of how likely they are to develop a specific health condition than considering any one risk factor one its own. The accuracy of polygenic risk scores becomes even more important when considering that these scores may be used to guide medication prescription or help patients make decisions about preventive surgery, such as a mastectomy.
In a study published in September, researchers used results from large genetics studies of people with European ancestry and data from the UK Biobank to calculate polygenic risk scores for breast and prostate cancer for people with African, East Asian, European and South Asian ancestry. They found that they could identify individuals at higher risk of breast and prostate cancer when they scaled the risk scores within each group, but the authors say this is only a temporary solution. Recruiting more diverse participants for genetics studies will lead to better cancer detection and prevent, they conclude.
Recent efforts to do just that are expected to make these scores more accurate in the future. Until then, some genetics companies are struggling to overcome the European bias in their tests.
Acknowledging the limitations of its polygenic risk score, Ambry Genetics said in April that it would stop offering the test until it could be recalibrated. The company launched the test, known as AmbryScore, in 2018.
"After careful consideration, we have decided to discontinue AmbryScore to help reduce disparities in access to genetic testing and to stay aligned with current guidelines," the company said in an email to customers. "Due to limited data across ethnic populations, most polygenic risk scores, including AmbryScore, have not been validated for use in patients of diverse backgrounds." (The company did not make a spokesperson available for an interview for this story.)
In September 2020, the National Comprehensive Cancer Network updated its guidelines to advise against the use of polygenic risk scores in routine patient care because of "significant limitations in interpretation." The nonprofit, which represents 31 major cancer cancers across the United States, said such scores could continue to be used experimentally in clinical trials, however.
Holly Pederson, director of Medical Breast Services at the Cleveland Clinic, says the realization that polygenic risk scores may not be accurate for all races and ethnicities is relatively recent. Pederson worked with Salt Lake City-based Myriad Genetics, a leading provider of genetic tests, to improve the accuracy of its polygenic risk score for breast cancer.
The company announced in August that it had recalibrated the test, called RiskScore, for women of all ancestries. Previously, Myriad did not offer its polygenic risk score to women who self-reported any ancestry other than sole European or Ashkenazi ancestry.
"Black women, while they have a similar rate of breast cancer to white women, if not lower, had twice as high of a polygenic risk score because the development and validation of the model was done in white populations," Pederson said of the old test. In other words, Myriad's old test would have shown that a Black woman had twice as high of a risk for breast cancer compared to the average woman even if she was at low or average risk.
To develop and validate the new score, Pederson and other researchers assessed data from more than 275,000 women, including more than 31,000 African American women and nearly 50,000 women of East Asian descent. They looked at 56 different genetic variants associated with ancestry and 93 associated with breast cancer. Interestingly, they found that at least 95% of the breast cancer variants were similar amongst the different ancestries.
The company says the resulting test is now more accurate for all women across the board, but Pederson cautions that it's still slightly less accurate for Black women.
"It's not only the lack of data from Black women that leads to inaccuracies and a lack of validation in these types of risk models, it's also the pure genomic diversity of Africa," she says, noting that Africa is the most genetically diverse continent on the planet. "We just need more data, not only in American Black women but in African women to really further characterize that continent."
Martin says it's problematic that such scores are most accurate for white people because they could further exacerbate health disparities in traditionally underserved groups, such as Black Americans. "If we were to set up really representative massive genetic studies, we would do a much better job at predicting genetic risk for everybody," she says.
Earlier this year, the National Institutes of Health awarded $38 million to researchers to improve the accuracy of polygenic risk scores in diverse populations. Researchers will create new genome datasets and pool information from existing ones in an effort to diversify the data that polygenic scores rely on. They plan to make these datasets available to other scientists to use.
"By having adequate representation, we can ensure that the results of a genetic test are widely applicable," Riordan says.
A robot server, controlled remotely by a disabled worker, delivers drinks to patrons at the DAWN cafe in Tokyo.
A sleek, four-foot tall white robot glides across a cafe storefront in Tokyo’s Nihonbashi district, holding a two-tiered serving tray full of tea sandwiches and pastries. The cafe’s patrons smile and say thanks as they take the tray—but it’s not the robot they’re thanking. Instead, the patrons are talking to the person controlling the robot—a restaurant employee who operates the avatar from the comfort of their home.
It’s a typical scene at DAWN, short for Diverse Avatar Working Network—a cafe that launched in Tokyo six years ago as an experimental pop-up and quickly became an overnight success. Today, the cafe is a permanent fixture in Nihonbashi, staffing roughly 60 remote workers who control the robots remotely and communicate to customers via a built-in microphone.
More than just a creative idea, however, DAWN is being hailed as a life-changing opportunity. The workers who control the robots remotely (known as “pilots”) all have disabilities that limit their ability to move around freely and travel outside their homes. Worldwide, an estimated 16 percent of the global population lives with a significant disability—and according to the World Health Organization, these disabilities give rise to other problems, such as exclusion from education, unemployment, and poverty.
These are all problems that Kentaro Yoshifuji, founder and CEO of Ory Laboratory, which supplies the robot servers at DAWN, is looking to correct. Yoshifuji, who was bedridden for several years in high school due to an undisclosed health problem, launched the company to help enable people who are house-bound or bedridden to more fully participate in society, as well as end the loneliness, isolation, and feelings of worthlessness that can sometimes go hand-in-hand with being disabled.
“It’s heartbreaking to think that [people with disabilities] feel they are a burden to society, or that they fear their families suffer by caring for them,” said Yoshifuji in an interview in 2020. “We are dedicating ourselves to providing workable, technology-based solutions. That is our purpose.”
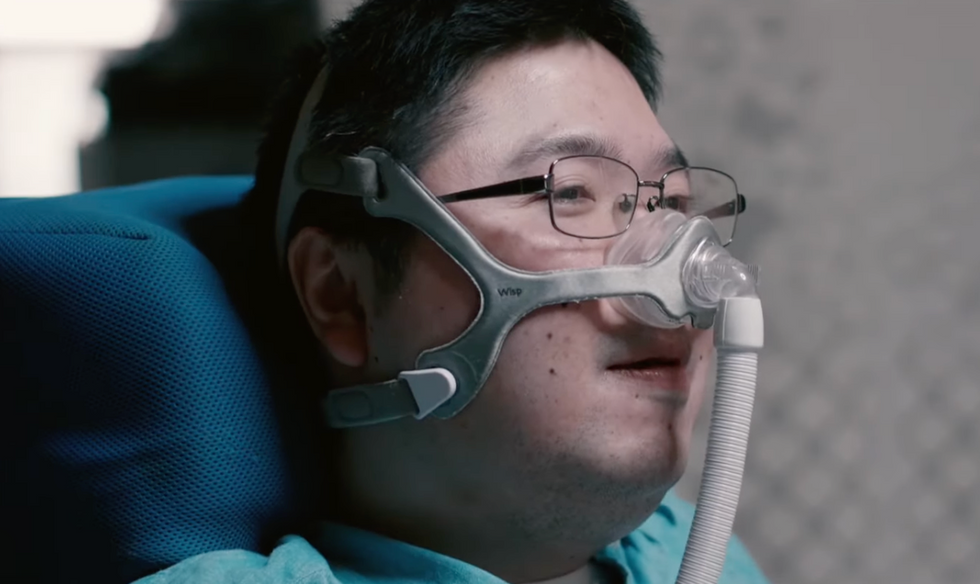
Shota, Kuwahara, a DAWN employee with muscular dystrophy, agrees. "There are many difficulties in my daily life, but I believe my life has a purpose and is not being wasted," he says. "Being useful, able to help other people, even feeling needed by others, is so motivational."
A woman receives a mammogram, which can detect the presence of tumors in a patient's breast.
When a patient is diagnosed with early-stage breast cancer, having surgery to remove the tumor is considered the standard of care. But what happens when a patient can’t have surgery?
Whether it’s due to high blood pressure, advanced age, heart issues, or other reasons, some breast cancer patients don’t qualify for a lumpectomy—one of the most common treatment options for early-stage breast cancer. A lumpectomy surgically removes the tumor while keeping the patient’s breast intact, while a mastectomy removes the entire breast and nearby lymph nodes.
Fortunately, a new technique called cryoablation is now available for breast cancer patients who either aren’t candidates for surgery or don’t feel comfortable undergoing a surgical procedure. With cryoablation, doctors use an ultrasound or CT scan to locate any tumors inside the patient’s breast. They then insert small, needle-like probes into the patient's breast which create an “ice ball” that surrounds the tumor and kills the cancer cells.
Cryoablation has been used for decades to treat cancers of the kidneys and liver—but only in the past few years have doctors been able to use the procedure to treat breast cancer patients. And while clinical trials have shown that cryoablation works for tumors smaller than 1.5 centimeters, a recent clinical trial at Memorial Sloan Kettering Cancer Center in New York has shown that it can work for larger tumors, too.
In this study, doctors performed cryoablation on patients whose tumors were, on average, 2.5 centimeters. The cryoablation procedure lasted for about 30 minutes, and patients were able to go home on the same day following treatment. Doctors then followed up with the patients after 16 months. In the follow-up, doctors found the recurrence rate for tumors after using cryoablation was only 10 percent.
For patients who don’t qualify for surgery, radiation and hormonal therapy is typically used to treat tumors. However, said Yolanda Brice, M.D., an interventional radiologist at Memorial Sloan Kettering Cancer Center, “when treated with only radiation and hormonal therapy, the tumors will eventually return.” Cryotherapy, Brice said, could be a more effective way to treat cancer for patients who can’t have surgery.
“The fact that we only saw a 10 percent recurrence rate in our study is incredibly promising,” she said.


