Meet the Psychologist Using Psychedelics to Treat Racial Trauma

Monnica Williams was stuck. The veteran psychologist wanted to conduct a study using psychedelics, but her university told her they didn't have the expertise to evaluate it via an institutional review board, which is responsible for providing ethical and regulatory oversight for research that involves human participants. Instead, they directed her to a hospital, whose reviewers turned it down, citing research of a banned substance as unethical.
"I said, 'We're not using illegal psilocybin, we're going through Health Canada,'" Williams said. Psilocybin was banned in Canada in 1974, but can now be obtained with an exemption from Health Canada, the federal government's health policy department. After learning this, the hospital review board told Williams they couldn't review her proposal because she's not affiliated with the hospital, after all.
It's all part of balancing bureaucracy with research goals for Williams, a leading expert on racial trauma and psychedelic medicine, as well as obsessive compulsive disorder (OCD), at the University of Ottawa. She's exploring the use of hallucinogenic substances like MDMA and psilocybin — commonly known as ecstasy and magic mushrooms, respectively — to help people of color address the psychological impacts of systemic racism. A prolific researcher, Williams also works as an expert witness, offering clinical evaluations for racial trauma cases.
Scientists have long known that psychedelics produce an altered state of consciousness and openness to new perspectives. For people with mental health conditions who haven't benefited from traditional therapy, psychedelics may be able to help them discover what's causing their pain or trauma, including racial trauma—the mental and emotional injury spurred by racial bias.
"Using psychedelics can not only bring these pain points to the surface for healing, but can reduce the anxiety or response to these memories and allow them to speak openly about them without the pain they bring," Williams says. Her research harnesses the potential of psychedelics to increase neuroplasticity, which includes the brain's ability to build new pathways.
"People of color are dealing with racism all the time, in large and small ways, and even dealing with racism in healthcare, even dealing with racism in therapy."
But she says therapists of color aren't automatically equipped to treat racial trauma. First, she notes, people of color are "vastly underrepresented in the mental health workforce." This is doubly true in psychedelic-assisted psychotherapy, in which a person is guided through a psychedelic session by a therapist or team of therapists, then processes the experience in subsequent therapy sessions.
"On top of that, the therapists of color are getting the same training that the white therapists are getting, so it's not even really guaranteed that they're going to be any better at helping a person that may have racial trauma emerging as part of their experience," she says.
In her own training to become a clinical psychologist at the University of Virginia, Williams says she was taught "how to be a great psychologist for white people." Yet even people of color, she argues, need specialized training to work with marginalized groups, particularly when it comes to MDMA, psilocybin and other psychedelics. Because these drugs can lower natural psychological defense mechanisms, Williams says, it's important for providers to be specially trained.
"People of color are dealing with racism all the time, in large and small ways, and even dealing with racism in healthcare, even dealing with racism in therapy. So [they] generally develop a lot of defenses and coping strategies to ward off racism so that they can function." she says. This is particularly true with psychedelic-assisted psychotherapy: "One possibility is that you're going to be stripped of your defenses, you're going to be vulnerable. And so you have to work with a therapist who is going to understand that and not enact more racism in their work with you."
Williams has struggled to find funding and institutional approval for research involving psychedelics, or funding for investigations into racial trauma or the impacts of conditions like OCD and post-traumatic stress disorder (PTSD) in people of color. With the bulk of her work focusing on OCD, she hoped to focus on people of color, but found there was little funding for that type of research. In 2020, that started to change as structural racism garnered more media attention.
After the killing of George Floyd, a 46-year-old Black man, by a white police officer in May 2020, Williams was flooded with media requests. "Usually, when something like that happens, I get contacted a lot for a couple of weeks, and it dies off. But after George Floyd, it just never did."
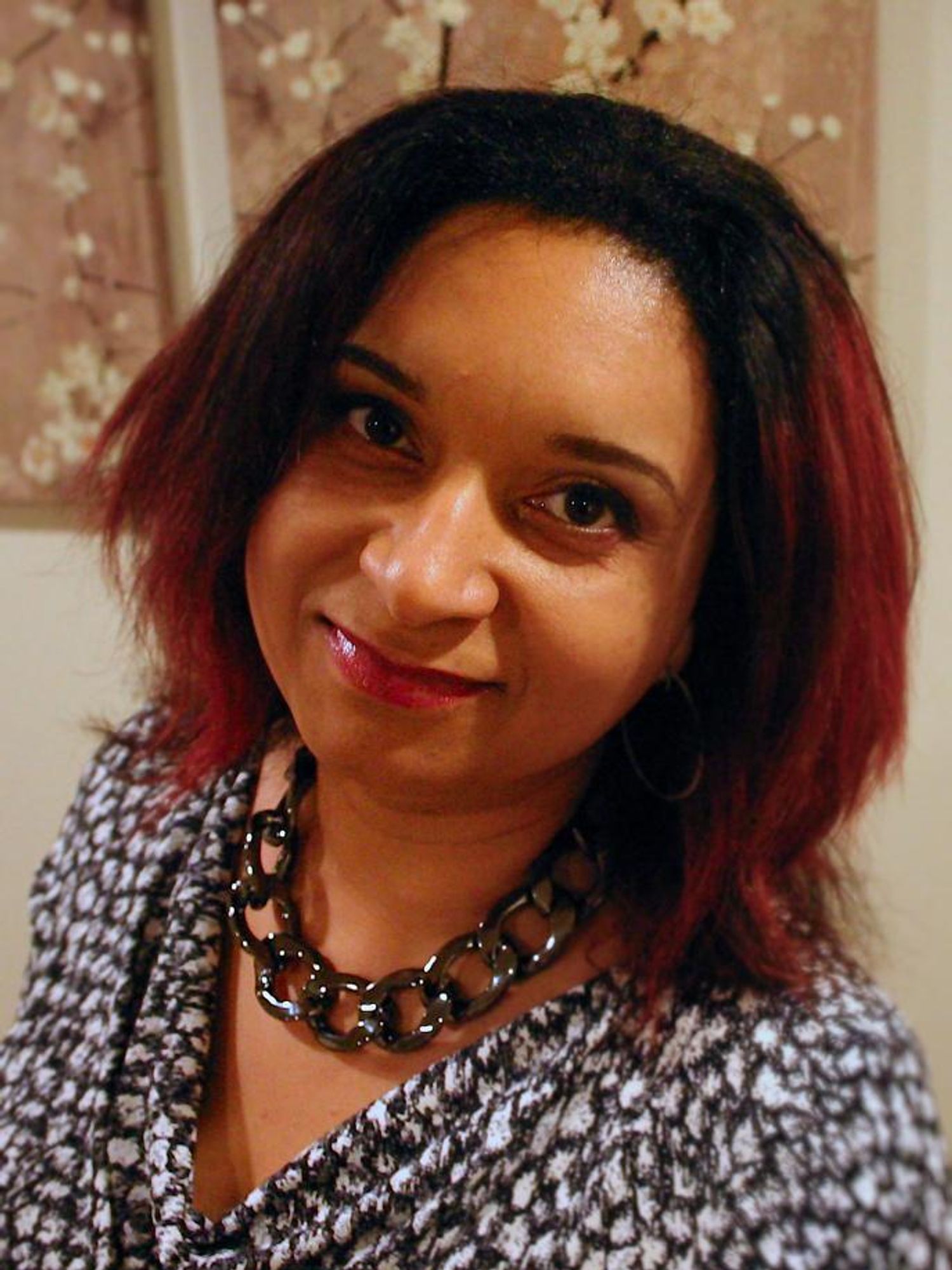
Monnica Williams, clinical psychologist at the University of Ottawa
Williams was no stranger to the questions that soon blazed across headlines: How can we mitigate microaggressions? How do race and ethnicity impact mental health? What terms should we use to discuss racial issues? What constitutes an ally, and why aren't there more of them? Why aren't there more people of color in academia, and so many other fields?
Now, she's hoping that the increased attention on racial justice will mean more acceptance for the kind of research she's doing.
In fact, Williams herself has used psychedelics in order to gain a better understanding of how to use them to treat racial trauma. In a study published in January, she and two other Black female psychotherapists took MDMA in a supervised setting, guided by a team of mental health practitioners who helped them process issues that came up as the session progressed. Williams, who was also the study's lead author, found that participants' experiences centered around processing and finding release from racial identities, and, in one case, of simply feeling wholly human without the burden of racial identity for the first time.
The purpose of the study was twofold: to understand how Black women react to psychedelics and to provide safe, firsthand, psychedelic experiences to Black mental health practitioners. One of the other study participants has since gone on to offer psychedelic-assisted psychotherapy to her own patients.
Psychedelic research, and psilocybin in particular, has become a hot topic of late, particularly after Oregon became the first state to legalize it for therapeutic use last November. A survey-based, observational study with 313 participants, published in 2020, paved the way for Williams' more recent MDMA experiments by describing improvements in depression, anxiety and racial trauma among people of color who had used LSD, psilocybin or MDMA in a non-research setting.
Williams and her team included only respondents who reported a moderate to strong psychoactive effect of past psychedelic consumption and believed these experiences provided "relief from the challenging effects of ethnic discrimination." Participants reported a memorable psychedelic experience as well as its acute and lasting effects, completing assessments of psychological insight, mystical experience and emotional challenges experienced during psychedelic experience, then describing their mental health — including depression, anxiety and trauma symptoms — before and after that experience.
Still, Williams says addressing racism is much more complex than treating racial trauma. "One of the questions I get asked a lot is, 'How can Black people cope with racism?' And I don't really like that question," she says. "I think it's important and I don't mind answering it, but I think the more important question is, how can we end racism? What can Black people do to stop racism that's happening to them and what can we do as a society to stop racism? And people aren't really asking this question."
A woman receives a mammogram, which can detect the presence of tumors in a patient's breast.
When a patient is diagnosed with early-stage breast cancer, having surgery to remove the tumor is considered the standard of care. But what happens when a patient can’t have surgery?
Whether it’s due to high blood pressure, advanced age, heart issues, or other reasons, some breast cancer patients don’t qualify for a lumpectomy—one of the most common treatment options for early-stage breast cancer. A lumpectomy surgically removes the tumor while keeping the patient’s breast intact, while a mastectomy removes the entire breast and nearby lymph nodes.
Fortunately, a new technique called cryoablation is now available for breast cancer patients who either aren’t candidates for surgery or don’t feel comfortable undergoing a surgical procedure. With cryoablation, doctors use an ultrasound or CT scan to locate any tumors inside the patient’s breast. They then insert small, needle-like probes into the patient's breast which create an “ice ball” that surrounds the tumor and kills the cancer cells.
Cryoablation has been used for decades to treat cancers of the kidneys and liver—but only in the past few years have doctors been able to use the procedure to treat breast cancer patients. And while clinical trials have shown that cryoablation works for tumors smaller than 1.5 centimeters, a recent clinical trial at Memorial Sloan Kettering Cancer Center in New York has shown that it can work for larger tumors, too.
In this study, doctors performed cryoablation on patients whose tumors were, on average, 2.5 centimeters. The cryoablation procedure lasted for about 30 minutes, and patients were able to go home on the same day following treatment. Doctors then followed up with the patients after 16 months. In the follow-up, doctors found the recurrence rate for tumors after using cryoablation was only 10 percent.
For patients who don’t qualify for surgery, radiation and hormonal therapy is typically used to treat tumors. However, said Yolanda Brice, M.D., an interventional radiologist at Memorial Sloan Kettering Cancer Center, “when treated with only radiation and hormonal therapy, the tumors will eventually return.” Cryotherapy, Brice said, could be a more effective way to treat cancer for patients who can’t have surgery.
“The fact that we only saw a 10 percent recurrence rate in our study is incredibly promising,” she said.
Urinary tract infections account for more than 8 million trips to the doctor each year.
Few things are more painful than a urinary tract infection (UTI). Common in men and women, these infections account for more than 8 million trips to the doctor each year and can cause an array of uncomfortable symptoms, from a burning feeling during urination to fever, vomiting, and chills. For an unlucky few, UTIs can be chronic—meaning that, despite treatment, they just keep coming back.
But new research, presented at the European Association of Urology (EAU) Congress in Paris this week, brings some hope to people who suffer from UTIs.
Clinicians from the Royal Berkshire Hospital presented the results of a long-term, nine-year clinical trial where 89 men and women who suffered from recurrent UTIs were given an oral vaccine called MV140, designed to prevent the infections. Every day for three months, the participants were given two sprays of the vaccine (flavored to taste like pineapple) and then followed over the course of nine years. Clinicians analyzed medical records and asked the study participants about symptoms to check whether any experienced UTIs or had any adverse reactions from taking the vaccine.
The results showed that across nine years, 48 of the participants (about 54%) remained completely infection-free. On average, the study participants remained infection free for 54.7 months—four and a half years.
“While we need to be pragmatic, this vaccine is a potential breakthrough in preventing UTIs and could offer a safe and effective alternative to conventional treatments,” said Gernot Bonita, Professor of Urology at the Alta Bro Medical Centre for Urology in Switzerland, who is also the EAU Chairman of Guidelines on Urological Infections.
The news comes as a relief not only for people who suffer chronic UTIs, but also to doctors who have seen an uptick in antibiotic-resistant UTIs in the past several years. Because UTIs usually require antibiotics, patients run the risk of developing a resistance to the antibiotics, making infections more difficult to treat. A preventative vaccine could mean less infections, less antibiotics, and less drug resistance overall.
“Many of our participants told us that having the vaccine restored their quality of life,” said Dr. Bob Yang, Consultant Urologist at the Royal Berkshire NHS Foundation Trust, who helped lead the research. “While we’re yet to look at the effect of this vaccine in different patient groups, this follow-up data suggests it could be a game-changer for UTI prevention if it’s offered widely, reducing the need for antibiotic treatments.”

