Are Brain Implants the Future of Treatment for Depression and Anxiety?
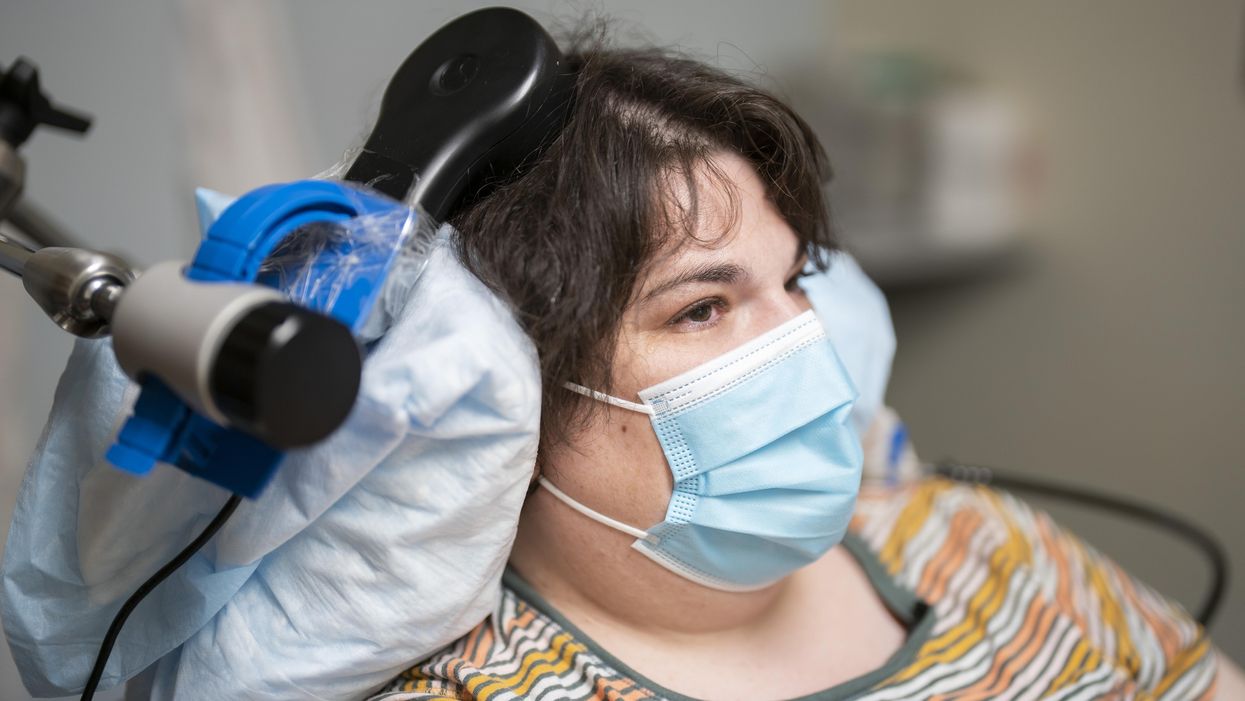
Sarah, clinical trial participant, at an appointment with Katherine Scangos, MD, PhD, at UCSF’s Langley Porter Psychiatric Institute.
When she woke up after a procedure involving drilling small holes in her skull, a woman suffering from chronic depression reported feeling “euphoric”. The holes were made to fit the wires that connected her brain with a matchbox-sized electrical implant; this would deliver up to 300 short-lived electricity bursts per day to specific parts of her brain.
Over a year later, Sarah, 36, says the brain implant has turned her life around. A sense of alertness and energy have replaced suicidal thoughts and feelings of despair, which had persisted despite antidepressants and electroconvulsive therapy. Sarah is the first person to have received a brain implant to treat depression, a breakthrough that happened during an experimental study published recently in Nature Medicine.
“What we did was use deep-brain stimulation (DBS), a technique used in the treatment of epilepsy,” says Andrew Krystal, professor of psychiatry at University of California, San Francisco (UCSF), and one of the study’s researchers. DBS typically involves implanting electrodes into specific areas of the brain to reduce seizures not controlled with medication or to remove the part of the brain that causes the seizures. Instead of choosing and stimulating a single brain site though, the UCSF team took a different approach.
They first used 10 electrodes to map Sarah’s brain activity, a phase that lasted 10 days, during which they developed a neural biomarker, a specific pattern of brain activity that indicated the onset of depression symptoms (in Sarah, this was detected in her amygdala, an almondlike structure located near the base of the brain). But they also saw that delivering a tiny burst of electricity to the patient’s ventral striatum, an area of the brain that sits in the center, above and behind the ears, dramatically improved these symptoms. What they had to do was outfit Sara’s brain with a DBS-device programmed to propagate small waves of electricity to the ventral striatum only when it discerned the pattern.
“We are not trying to take away normal responses to the world. We are just trying to eliminate this one thing, which is depression, which impedes patients’ ability to function and deal with normal stuff.”
“It was a personalized treatment not only in where to stimulate, but when to stimulate,” Krystal says. Sarah’s depression translated to low amounts of energy, loss of pleasure and interest in life, and feelings of sluggishness. Those symptoms went away when scientists stimulated her ventral capsule area. When the same area was manipulated by electricity when Sarah’s symptoms “were not there” though, she was feeling more energetic, but this sudden flush of energy soon gave way to feelings of overstimulation and anxiety. “This is a very tangible illustration of why it's best to simulate only when you need it,” says Krystal.
We have the tendency to lump together depression symptoms, but, in reality, they are quite diverse; some people feel sad and lethargic, others stay up all night; some overeat, others don’t eat at all. “This happens because people have different underlying dysfunctions in different parts of their brain. Our approach is targeting the specific brain circuit that modulates different kinds of symptoms. Simply, where we stimulate depends on the specific set of problems a person has,” Krystal says. Such tailormade brain stimulation for patients with long-term, drug-resistant depression, which would be easy to use at home, could be transformative, the UCSF researcher concludes.
In the U.S., 12.7 percent of the population is on antidepressants. Almost exactly the same percentage of Australians–12.5–take similar drugs every day. With 13 percent of its population being on antidepressants, Iceland is the world’s highest antidepressant consumer. And quite away from Scandinavia, the Southern European country of Portugal is the world’s third strongest market for corresponding medication.
By 2020, nearly 15.5 million people had been consuming antidepressants for a time period exceeding five years. Between 40 and 60 percent of them saw improvements. “For those people, it was absolutely what they needed, whether that was increased serotonin, or increased norepinephrine or increased dopamine, ” says Frank Anderson, a psychiatrist who has been administering antidepressants in his private practice “for a long time”, and author of Transcending Trauma, a book about resolving complex and dissociative trauma.
Yet the UCSF study brings to the mental health field a specificity it has long lacked. “A lot of the traditional medications only really work on six neurotransmitters, when there are over 100 neurotransmitters in the brain,” Anderson says. Drugs are changing the chemistry of a single system in the brain, but brain stimulation is essentially changing the very architecture of the brain, says James Giordano, professor of neurology and biochemistry at Georgetown University Medical Center in Washington and a neuroethicist. It is a far more elegant approach to treating brain disorders, with the potential to prove a lifesaver for the 40 to 50 percent of patients who see no benefits at all with antidepressants, Giordano says. It is neurofeedback, on steroids, adds Anderson. But it comes with certain risks.
Even if the device generating the brain stimulation sits outside the skull and could be easily used at home, the whole process still involves neurosurgery. While the sophistication and precision of brain surgeries has significantly improved over the last years, says Giordano, they always carry risks, such as an allergic reaction to anesthesia, bleeding in the brain, infection at the wound site, blood clots, even coma. Non-invasive brain stimulation (NIBS), a technology currently being developed by the Defense Advanced Research Projects Agency (DARPA), could potentially tackle this. Patients could wear a cap, helmet, or visor that transmits electrical signals from the brain to a computer system and back, in a brain-computer interface that would not need surgery.
“This could counter the implantation of hardware into the brain and body, around which there is also a lot of public hesitance,” says Giordano, who is working on such techniques at DARPA.
Embedding a chip in your head is one of the finest examples of biohacking, an umbrella word for all the practices aimed at hacking one’s body and brain to enhance performance –a citizen do-it-yourself biology. It is also a word charged enough to set off a public backlash. Large segments of the population will simply refuse to allow that level of invasiveness in their heads, says Laura Cabrera, an associate professor of neuroethics at the Center for Neural Engineering, Department of Engineering Science and Mechanics at Penn State University. Cabrera urges caution when it comes to DBS’s potential.
“We've been using it for Parkinson's for over two decades, hoping that now that they get DBS, patients will get off medications. But people have continued taking their drugs, even increasing them,” she says. What the UCSF found is a proof of concept that DBS worked in one depressed person, but there’s a long way ahead until we can confidently say this finding is generalizable to a large group of patients. Besides, as a society, we are not there yet, says Cabrera. “Most people, at least in my research, say they don't want to have things in their brain,” she says. But what could really go wrong if we biohacked our own brains anyway?
In 2014, a man who had received a deep brain implant for a movement disorder started developing an affection for Johnny Cash’s music when he had previously been an avid country music fan. Many protested that the chip had tampered with his personality. Could sparking the brain with electricity generated by a chip outside it put an end to our individuality, messing with our musical preferences, unique quirks, our deeper sense of ego?
“What we found is that when you stimulate a region, you affect people’s moods, their energies,” says Krystal. You are neither changing their personality nor creating creatures of eternal happiness, he says. “’Being on a phone call would generally be a setting that would normally trigger symptoms of depression in me,’” Krystal reports his patient telling him. ‘I now know bad things happen, but am not affected by them in the same way. They don’t trigger the depression.’” Of the research, Krystal continues: “We are not trying to take away normal responses to the world. We are just trying to eliminate this one thing, which is depression, which impedes patients’ ability to function and deal with normal stuff.”
Yet even change itself shouldn't be seen as threatening, especially if the patient had probably desired it in the first place. “The intent of therapy in psychiatric disorders is to change the personality, because a psychiatric disorder by definition is a disorder of personality,” says Cabrera. A person in therapy wants to restore the lost sense of “normal self”. And as for this restoration altering your original taste in music, Cabrera says we are talking about rarities, extremely scarce phenomena that are possible with medication as well.
Maybe it is the allure of dystopian sci-fi films: people have a tendency to worry about dark forces that will spread malice across the world when the line between human and machine has blurred. Such mind-control through DBS would probably require a decent leap of logic with the tools science has--at least to this day. “This would require an understanding of the parameters of brain stimulation we still don't have,” says Cabrera. Still, brain implants are not fully corrupt-proof.
“Hackers could shut off the device or change the parameters of the patient's neurological function enhancing symptoms or creating harmful side-effects,” says Giordano.
There are risks, but also failsafe ways to tackle them, adds Anderson. “Just like medications are not permanent, we could ensure the implants are used for a specific period of time,” he says. And just like people go in for checkups when they are under medication, they could periodically get their personal brain implants checked to see if they have been altered or not, he continues. “It is what my research group refers to as biosecurity by design,” says Giordano. “It is important that we proactively design systems that cannot be corrupted.”
Two weeks after receiving the implant, Sarah scored 14 out of 54 on the Montgomery-Åsberg Depression Rating Scale, a ten-item questionnaire psychiatrists use to measure the severity of depressive episodes. She had initially scored 36. Today she scores under 10. She would have had to wait between four and eight weeks to see positive results had she taken the antidepressant road, says Krystal.
He and his team have enrolled two other patients in the trials and hope to add nine more. They already have some preliminary evidence that there's another place that works better in the brain of another patient, because that specific patient had been experiencing more anxiety as opposed to despondency. Almost certainly, we will have different biomarkers for different people, and brain stimulation will be tailored to a person’s unique situation, says Krystal. “Each brain is different, just like each face is different.”
A woman receives a mammogram, which can detect the presence of tumors in a patient's breast.
When a patient is diagnosed with early-stage breast cancer, having surgery to remove the tumor is considered the standard of care. But what happens when a patient can’t have surgery?
Whether it’s due to high blood pressure, advanced age, heart issues, or other reasons, some breast cancer patients don’t qualify for a lumpectomy—one of the most common treatment options for early-stage breast cancer. A lumpectomy surgically removes the tumor while keeping the patient’s breast intact, while a mastectomy removes the entire breast and nearby lymph nodes.
Fortunately, a new technique called cryoablation is now available for breast cancer patients who either aren’t candidates for surgery or don’t feel comfortable undergoing a surgical procedure. With cryoablation, doctors use an ultrasound or CT scan to locate any tumors inside the patient’s breast. They then insert small, needle-like probes into the patient's breast which create an “ice ball” that surrounds the tumor and kills the cancer cells.
Cryoablation has been used for decades to treat cancers of the kidneys and liver—but only in the past few years have doctors been able to use the procedure to treat breast cancer patients. And while clinical trials have shown that cryoablation works for tumors smaller than 1.5 centimeters, a recent clinical trial at Memorial Sloan Kettering Cancer Center in New York has shown that it can work for larger tumors, too.
In this study, doctors performed cryoablation on patients whose tumors were, on average, 2.5 centimeters. The cryoablation procedure lasted for about 30 minutes, and patients were able to go home on the same day following treatment. Doctors then followed up with the patients after 16 months. In the follow-up, doctors found the recurrence rate for tumors after using cryoablation was only 10 percent.
For patients who don’t qualify for surgery, radiation and hormonal therapy is typically used to treat tumors. However, said Yolanda Brice, M.D., an interventional radiologist at Memorial Sloan Kettering Cancer Center, “when treated with only radiation and hormonal therapy, the tumors will eventually return.” Cryotherapy, Brice said, could be a more effective way to treat cancer for patients who can’t have surgery.
“The fact that we only saw a 10 percent recurrence rate in our study is incredibly promising,” she said.
Urinary tract infections account for more than 8 million trips to the doctor each year.
Few things are more painful than a urinary tract infection (UTI). Common in men and women, these infections account for more than 8 million trips to the doctor each year and can cause an array of uncomfortable symptoms, from a burning feeling during urination to fever, vomiting, and chills. For an unlucky few, UTIs can be chronic—meaning that, despite treatment, they just keep coming back.
But new research, presented at the European Association of Urology (EAU) Congress in Paris this week, brings some hope to people who suffer from UTIs.
Clinicians from the Royal Berkshire Hospital presented the results of a long-term, nine-year clinical trial where 89 men and women who suffered from recurrent UTIs were given an oral vaccine called MV140, designed to prevent the infections. Every day for three months, the participants were given two sprays of the vaccine (flavored to taste like pineapple) and then followed over the course of nine years. Clinicians analyzed medical records and asked the study participants about symptoms to check whether any experienced UTIs or had any adverse reactions from taking the vaccine.
The results showed that across nine years, 48 of the participants (about 54%) remained completely infection-free. On average, the study participants remained infection free for 54.7 months—four and a half years.
“While we need to be pragmatic, this vaccine is a potential breakthrough in preventing UTIs and could offer a safe and effective alternative to conventional treatments,” said Gernot Bonita, Professor of Urology at the Alta Bro Medical Centre for Urology in Switzerland, who is also the EAU Chairman of Guidelines on Urological Infections.
The news comes as a relief not only for people who suffer chronic UTIs, but also to doctors who have seen an uptick in antibiotic-resistant UTIs in the past several years. Because UTIs usually require antibiotics, patients run the risk of developing a resistance to the antibiotics, making infections more difficult to treat. A preventative vaccine could mean less infections, less antibiotics, and less drug resistance overall.
“Many of our participants told us that having the vaccine restored their quality of life,” said Dr. Bob Yang, Consultant Urologist at the Royal Berkshire NHS Foundation Trust, who helped lead the research. “While we’re yet to look at the effect of this vaccine in different patient groups, this follow-up data suggests it could be a game-changer for UTI prevention if it’s offered widely, reducing the need for antibiotic treatments.”

